 The Patton-Reeder indicator is used here in the form of a. 
molecule which creates a complex with a metal ion, bonding through six coordination sites. If possible, dry 5 g of the disodium salt of EDTA. This page contains answers to puzzle Ion bonding capacity measure. 10 mL and 100 mL measuring cylinders burette and stand Solutions Needed EDTA: ethylenediaminetetraacetic acid 0.025 mol L1 solution. It is often used for inorganic anions (e.g., chloride, nitrate, and sulfate) and inorganic cations (e.g., lithium, sodium, and potassium). This technique is used to analyze ionic substances. The answers are divided into several pages to keep it clear. A second sub-category of liquid chromatography is known as ion-exchange chromatography. Become a master crossword solver while having tons of fun, and all for free! A cation with a \(2 \) charge will make a stronger ionic bond than a cation with a \(1 \) charge. The strength of the ionic bond is directly dependent upon the quantity of the charges and inversely dependent on the distance between the charged particles. Increase your vocabulary and general knowledge. An ionic bond is the electrostatic force that holds ions together in an ionic compound. Give your brain some exercise and solve your way through brilliant crosswords published every day! Choose from a range of topics like Movies, Sports, Technology, Games, History, Architecture and more!Īccess to hundreds of puzzles, right on your Android device, so play or review your crosswords when you want, wherever you want! Hence, the valence of carbon is 4.Hello everyone! Thank you visiting our website, here you will be able to find all the answers for Daily Themed Crossword Game (DTC).ĭaily Themed Crossword is the new wonderful word game developed by PlaySimple Games, known by his best puzzle word games on the android and apple store.Ī fun crossword game with each day connected to a different theme. For example, in methane \ molecules, carbon atoms form a total of four bonds with four hydrogen atoms. Note: The valency of an atom represents the number of bonds it can form with other atoms. The protein binding capacity increases with an increased degree of. So the combining capacity of an element is known as its valency. The principle for protein adsorption to HIC media is complementary to ion exchange. To attain stability, magnesium loses two valence electrons and completes its octet. On the other hand, magnesium has two electrons. To attain stability, sodium loses one valence electron and completes its octet. For example, sodium has only one electron in its valence shell. In this way, it attains stability.įor metals, you can calculate the valence from its valence shell electronic configuration. Provide proper financial statements and reports Work with a construction CPA so you know which reports the surety will be looking at, and make sure they are presented professionally. To attain stability, oxygen gains two valence electrons and completes its octet. There are four primary ways for contractors to boost their bonding capacity. Similarly, oxygen has 6 valence electrons. To attain stability, chlorine gains one valence electron and completes its octet. 
The outermost shell is also called the valence shell. (WHC) (or water-binding capacity, or water-absorption capacity) is a measure of. The valence electrons are the number of electrons present in the outermost shell. CHEMICAL AND PHYSICAL CHARACTERISTICS OF MEAT Water-Holding Capacity. The combining capacity of an element is known as its valency.įor a nonmetal, you can calculate its valence by subtracting the number of valence electrons from 8. You can easily improve your search by specifying the number of letters in the answer. Below are all possible answers to this clue ordered by its rank. We found 20 possible solutions for this clue. with 7 letters was last seen on the January 01, 2013. The combining capacity of an element is dependent upon the number of electrons needed by an atom to achieve stable electronic configuration. The crossword clue Bonding capacity measure. 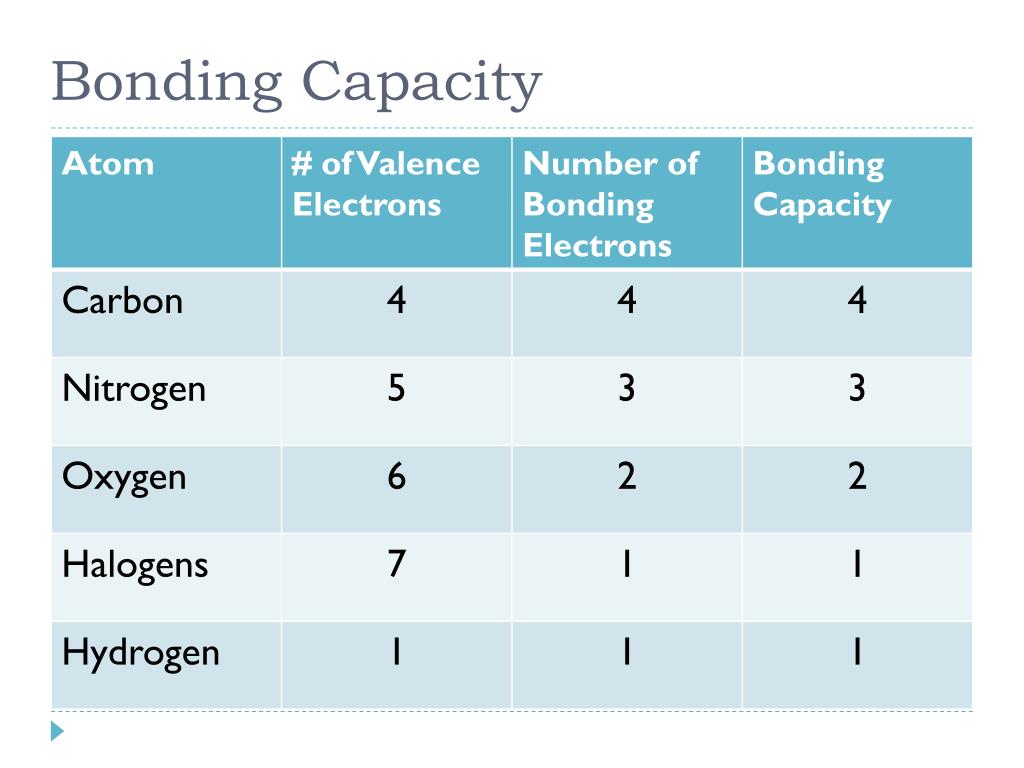
Thus, if you count the number of bonds around the central atom, you can determine its combining capacity. The bond energy for a diatomic molecule, D X Y, is defined as the standard enthalpy change for the endothermic reaction: (9.4.1) X Y ( g) X ( g) Y ( g) D X Y H °. We found 20 possible solutions for this clue. The energy required to break a specific covalent bond in one mole of gaseous molecules is called the bond energy or the bond dissociation energy. Hint: The combining capacity of an element will tell you the number of atoms that are bonded to the given atom. The crossword clue Bonding capacity measure with 7 letters was last seen on the January 01, 2013. 
0 Comments
Leave a Reply. |



 RSS Feed
RSS Feed
